Product Center
Navigation
Product Categories
![Organic And lnorganic Compound Fertilizer Organic And lnorganic Compound Fertilizer]() Organic And lnorganic Compound FertilizerAmino acid organic fertilizer is derived or fermented from natural organic materials, rich in amino acids and various nutrients essential for plant growth.
Organic And lnorganic Compound FertilizerAmino acid organic fertilizer is derived or fermented from natural organic materials, rich in amino acids and various nutrients essential for plant growth.![Foliar Fertilizer Foliar Fertilizer]() Foliar FertilizerAmino acid water soluble fertilizer is a kind of fertilizer which can be directly absorbed and utilized by crops. It can promote photosynthesis and the formation of chlorophyll, and significantly promote and activate physiological and biochemical processes such as oxide activity, enzyme activity, seed germination, nutrient absorption, root growth and development.
Foliar FertilizerAmino acid water soluble fertilizer is a kind of fertilizer which can be directly absorbed and utilized by crops. It can promote photosynthesis and the formation of chlorophyll, and significantly promote and activate physiological and biochemical processes such as oxide activity, enzyme activity, seed germination, nutrient absorption, root growth and development.![Calcium Ammonium Nitrate Calcium Ammonium Nitrate]() Calcium Ammonium NitrateSuitable for a variety of soil and crops, widely used in greenhouse and field planting food crops, economic crops, flowers, fruit trees, vegetables and so on. The nitrate nitrogen in calcium ammonium nitrate can be quickly dissolved in water and directly absorbed by plants. Suitable for base fertilizer, seed fertilizer and topdressing fertilizer.
Calcium Ammonium NitrateSuitable for a variety of soil and crops, widely used in greenhouse and field planting food crops, economic crops, flowers, fruit trees, vegetables and so on. The nitrate nitrogen in calcium ammonium nitrate can be quickly dissolved in water and directly absorbed by plants. Suitable for base fertilizer, seed fertilizer and topdressing fertilizer.
Send Us A Message
Strong Alkalinity – Highly effective for chemical reactions, cleaning, and neutralization processes.
Multiple Solubility Options – Readily dissolves in water, methanol, and ethanol for flexible usage.
Moisture and Gas Absorption – Deliquescent nature enables absorption of water vapor and acidic gases like CO₂.
PRODUCT PARAMETERS
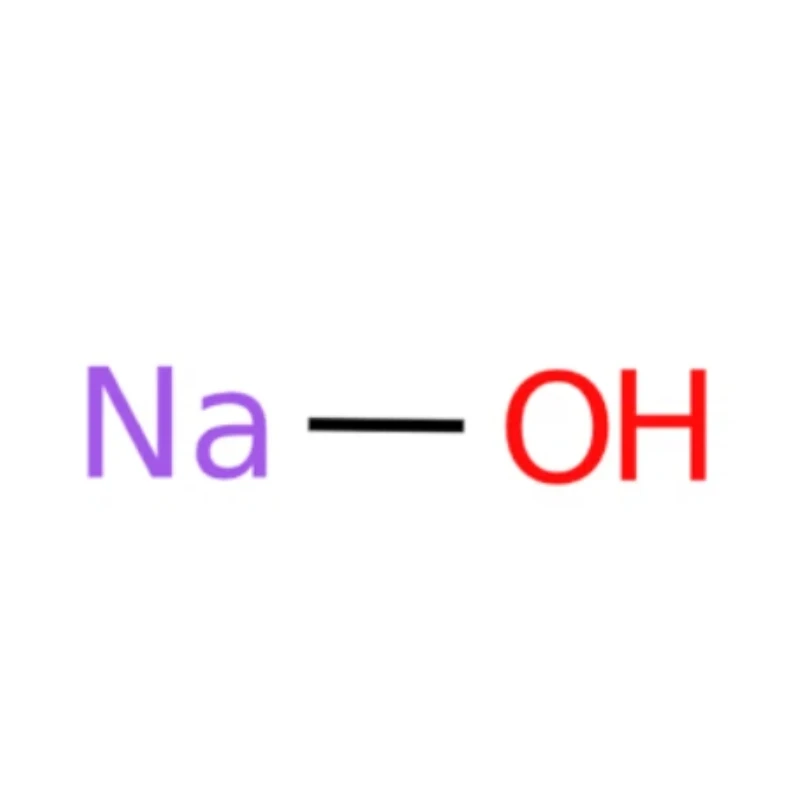
Sodium hydroxide Chemical Properties
Safety Information
Sodium hydroxide has a broad range of applications. In chemical experiments, it serves not only as a reagent but also as an alkaline desiccant due to its strong hygroscopic properties. Caustic soda plays a vital role in the national economy, with numerous industrial sectors relying on it. The largest consumer is the chemical manufacturing industry, followed by paper production, aluminum and tungsten smelting, viscose fiber manufacturing, and soap production. It is also used in the manufacture of dyes, plastics, pharmaceuticals, organic intermediates, reclaimed rubber, metal sodium electrolysis, water treatment, and inorganic salt production such as borax, chromium salts, manganates, and phosphates.
Industrial sodium hydroxide must meet the requirements of national standard GB209-2006;
Industrial ion-exchange membrane sodium hydroxide should comply with GB/T11199-89;
Sodium hydroxide for chemical fiber should meet GB11212-89;
Food-grade sodium hydroxide must conform to GB5175-85.
In industrial contexts, sodium hydroxide is commonly referred to as caustic soda, a name derived from its ability to cause severe skin burns upon contact with concentrated solutions. It dissolves proteins, making alkali burns slower to heal than acid burns. A 0.02% solution instilled into rabbit eyes can damage the corneal epithelium. Toxicity data include: mouse intraperitoneal LD50 of 40 mg/kg and rabbit oral LDLo of 500 mg/kg. Dust can irritate the eyes and respiratory tract, corrode the nasal septum, and damage skin—especially mucous membranes—producing soft scabs that may penetrate deeply and leave permanent scars.
PRODUCT DISPLAY
FACTORY DISPLAY
FAQ
How can I obtain more information or begin business discussions?
What are the main advantages of partnering with your company?
Resource Network: Stable and high-quality domestic supply chain.
Reliable Quality: Comprehensive quality control processes.
Efficient Service: Experienced team providing one-stop service, with smooth and efficient communication.
Guaranteed Reputation: Based on integrity, pursuing long-term win-win cooperation.
Do you handle export logistics and customs clearance?
What trade terms and payment methods do you support?
What is your typical delivery time?
How to inquire about prices and obtain samples?
How do you guarantee product quality?
Source control: We carefully select our suppliers to ensure they have reliable production qualifications and stable quality systems.
Process control: We can arrange factory inspections or third-party testing to ensure products meet contract standards and relevant international requirements.
Complete documentation: We provide the necessary quality certification documents, such as COA (Certificate of Analysis).
Can you provide customized products or meet special specifications?
What are your main exported chemical products?
What are the company's main markets and customer groups?
What kind of company is Jinan Yifu Chemical Co., Ltd.?
PLEASE CONTACT US
PRODUCT RECOMMENDATION